
Practitioner's Corner
J Investig Allergol Clin Immunol 2019; Vol. 29(5): 378-398
© 2019 Esmon Publicidad
written informed consent to participate. The study was
conducted following the principles of the Declaration of
Helsinki and approved by Fundación Jiménez Díaz Ethics
Committee. All selected patients had a confirmed diagnosis
of asthma with >12% improvement in FEV
1
15 minutes
after inhaling salbutamol (400 μg) or methacholine airway
hyperresponsiveness (PC
20
methacholine <16 mg/mL) [9].
They also had moderate persistent asthma and were being
treated with a combination of inhaled corticosteroids/long-
acting ß-agonists at medium doses (400 µg of budesonide and
12 µg of formoterol fumarate dihydrate daily or equivalent).
No change was made in the treatment received for asthma
during the study period, ie, from baseline to the follow-up
visits. Serum was obtained by centrifugation and stored at
–80ºC before analysis for no more than 2 years.
Serum miRNAs were extracted using the miRCURY
RNA Isolation Kit-Biofluids (Qiagen) and retrotranscribed
to cDNA using the Universal cDNA Synthesis kit II (Qiagen)
following the manufacturer’s instructions. Synthetic spike-ins
were added during the RNA extraction (Sp2, Sp4, and Sp5)
and reverse transcription (Sp6) processes to ensure appropriate
extraction and cDNA synthesis. miRNA expression was
evaluated using quantitative polymerase chain reaction as
previously described [10] at baseline and 6-12 months later
at follow-up visits.
The miRNAs analyzed were miR-320-a, miR-144-5p,
miR-1246, miR-21-5p, and miR-185-5p. These miRNAs
were selected because we previously found that their profile in
eosinophils can be used as a serum biomarker of asthma [10].
MiR-191-5p was measured as the endogenous control, and
Sp2, Sp4, Sp5, and Sp6 were measured to ensure correct
extraction and reverse transcription.
The statistical analysis was carried out using the GraphPad
Instat program. The
t
test was performed for normally
distributed samples (those meeting a Gaussian distribution)
and the Mann-Whitney test for non-normally distributed
samples. Paired tests were performed to compare baseline
data with follow-up data.
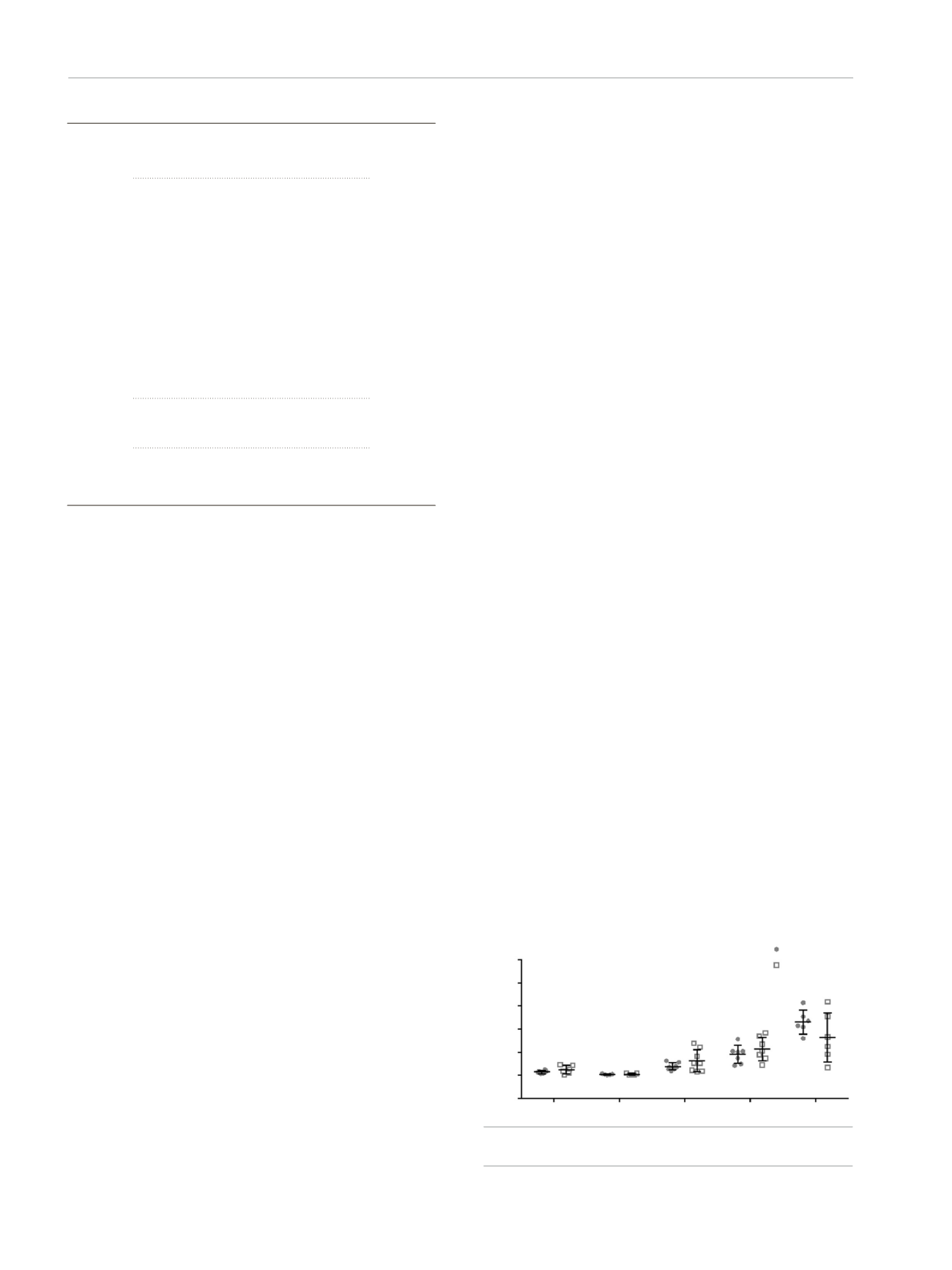
Asthma was stable over time in terms of the mean (SD)
Asthma Control Test score (21.1 [3.7] vs 20.8 [3.1]) and lung
function (FEV
1
%, 97.7 [12.9] vs 97.5 [13.9]). In addition
to the stable clinical parameters, no statistically significant
differences were found between the results obtained in
asthmatics at baseline and follow-up visits for any of the
Stability of Asthma Control Implies No Changes in
microRNAs Expression
Rial MJ
1,3
*, Rodrigo-Muñoz JM
2,3
*, Sastre B
2,3
, Sastre J
1,3
, del
Pozo V
2,3
1
Department of Allergy, Hospital Universitario Fundación
Jimenez Diaz, Instituto de Investigación Sanitaria Fundacion
Jimenez Diaz (IIS-FJD, UAM), Madrid, Spain
2
Department of Immunology, Instituto de Investigación Sanitaria
Hospital Universitario Fundacion Jimenez Diaz. (IIS-FJD, UAM),
Madrid, Spain
3
CIBER de Enfermedades Respiratorias (CIBERES), Instituto de
Salud Carlos III, Spain
*
These authors contributed equally to the manuscript.
J Investig Allergol Clin Immunol 2019; Vol. 29(5): 388-389
doi: 10.18176/jiaci.0410
Key words:
Asthma. miRNA. Biomarkers. Eosinophils. Exosomes.
Palabras clave:
Asma. miRNA. Biomarcadores. Eosinófilos. Exosomas.
Asthma is a chronic disease that affects 4.3% of the
population worldwide [1]. Pulmonary function tests and
bronchial provocation tests are still the gold standard in
diagnosing and assessing the severity of respiratory diseases;
however, they are not able to differentiate between the clinical
phenotypes responsible for specific manifestations. An ideal
biomarker must be measurable with minimal invasiveness,
be specific and sensitive, and be able to be detected quickly
and accurately. In this context, microRNAs (miRNAs)
present in body fluids have been reported to meet several
of these criteria and are used as diagnostic markers in
many areas [2-4]. Eosinophils, which play a key role in the
pathogenesis of asthma, have the ability to secrete exosomes.
These structures contain miRNAs, which are single-stranded
RNA sequences (around 19-22 nucleotides) that do not code
for proteins with crucial functions in the development and
maintenance of the pathogenic mechanisms of asthma, but
may instead be implicated in the pathophysiology of asthma
by regulating the translation of proteins related to asthma
processes [5]. miRNAs can be encapsulated in exosomes or
bound to proteins in biofluids; in both cases, they are highly
resistant to degradation by RNases [6,7].
Released exosomes and the miRNAs inside them have
been found in serum. However, neither the precise role of
miRNAs in asthma nor their stability in the same patient over
time has been fully defined [3-5].
In order to establish whether expression of miRNA in
clinically stable asthma patients remains steady over time, we
selected 20 asthmatic patients from a national cohort (MEGA
project) [8]. These patients were recruited randomly in the
Allergy Department of Fundación Jiménez Díaz University
Hospital, Madrid, Spain. Clinical and epidemiological
characteristics are shown in the Supplementary Material.
Patients received all necessary information and gave their
Figure.
Expression of miRNAs by qPCR at baseline and at follow-up visits.
Relative expression (2
-ΔCt
) is shown as mean (SD).
25
20
15
10
5
0
-5
miR-21-5p
miR-320a miR-185-5p
miR-144-5p
miR-1246
Relative miRNA Expression (2
-ΔCt
)
Baseline
Follow-up
388









